
Source of data: CRC Handbook of Chemistry and Physics,84th Edition (2004). An enthalpyentropy chart, also known as the HS chart or Mollier diagram, plots the total heat against entropy, describing the enthalpy of a thermodynamic. Standard thermodynamic Quantities for Chemical Substances at 25☌. There might be decreases in freedom in the rest of the universe, but the sum of the increase and decrease must result in a net increase.\) A new look at entropy and the life table, Demography, Springer Population Association of America (PAA), vol. Reaction thermochemistry data for over 8000 reactions. The freedom in that part of the universe may increase with no change in the freedom of the rest of the universe. Entropy Phase transition enthalpies and temperatures Vapor pressure. Statistical Entropy - Mass, Energy, and Freedom The energy or the mass of a part of the universe may increase or decrease, but only if there is a corresponding decrease or increase somewhere else in the universe.Compressed liquid: from tables Saturated liquid (sf): from tables. Qualitatively, entropy is simply a measure how much the energy of atoms and molecules become more spread out in a process and can be defined in terms of statistical probabilities of a system or in terms of the other thermodynamic quantities. The value of entropy at a specified state is determined just like any other property. Statistical Entropy Entropy is a state function that is often erroneously referred to as the 'state of disorder' of a system.In this context, the term usually refers to the Shannon entropy, which quantifies the expected value of the messages information. In information theory, entropy is a measure of the uncertainty in a random variable. 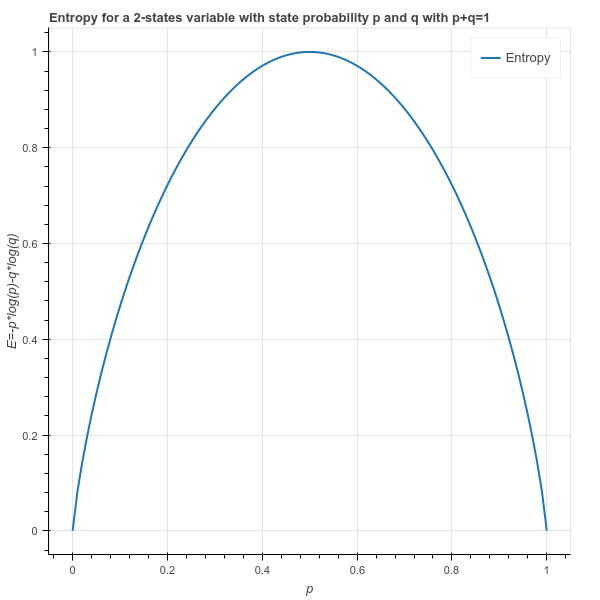
Phase Change, gas expansions, dilution, colligative properties and osmosis. This online calculator computes Shannon entropy for a given event probability table and for a given message. Simple Entropy Changes - Examples Several Examples are given to demonstrate how the statistical definition of entropy and the 2nd law can be applied. Contrary to previous claims, it is theoretically possible, and has been observed empirically, for life tables to have entropy values greater than unity.An example of numerical reduction of a molecule in the form of fractional Shannon entropy. Composition of air using nitrogen, argon, oxygen, and carbon dioxide as constituents. An example of numerical reduction of a molecule in the form of Shannon entropy. A microstate is one of the huge number of different accessible arrangements of the molecules' motional energy* for a particular macrostate. 362 9.1 Representative Tables of Thermodynamic Properties of Air. Instead, they are two very different ways of looking at a system. The entropy (very common in Information Theory) characterizes the (im)purityof an arbitrary collection of examples Information Gain is the expected reduction in entropy caused by partitioning the examples according to a given attribute Dip. 4.1 How to understand Shannon’s information entropy Entropy measures the degree of our lack of information about a system. We have changed their notation to avoid confusion. Unfortunately, in the information theory, the symbol for entropy is Hand the constant k B is absent. Microstates Dictionaries define “macro” as large and “micro” as very small but a macrostate and a microstate in thermodynamics aren't just definitions of big and little sizes of chemical systems. This expression is called Shannon Entropy or Information Entropy. 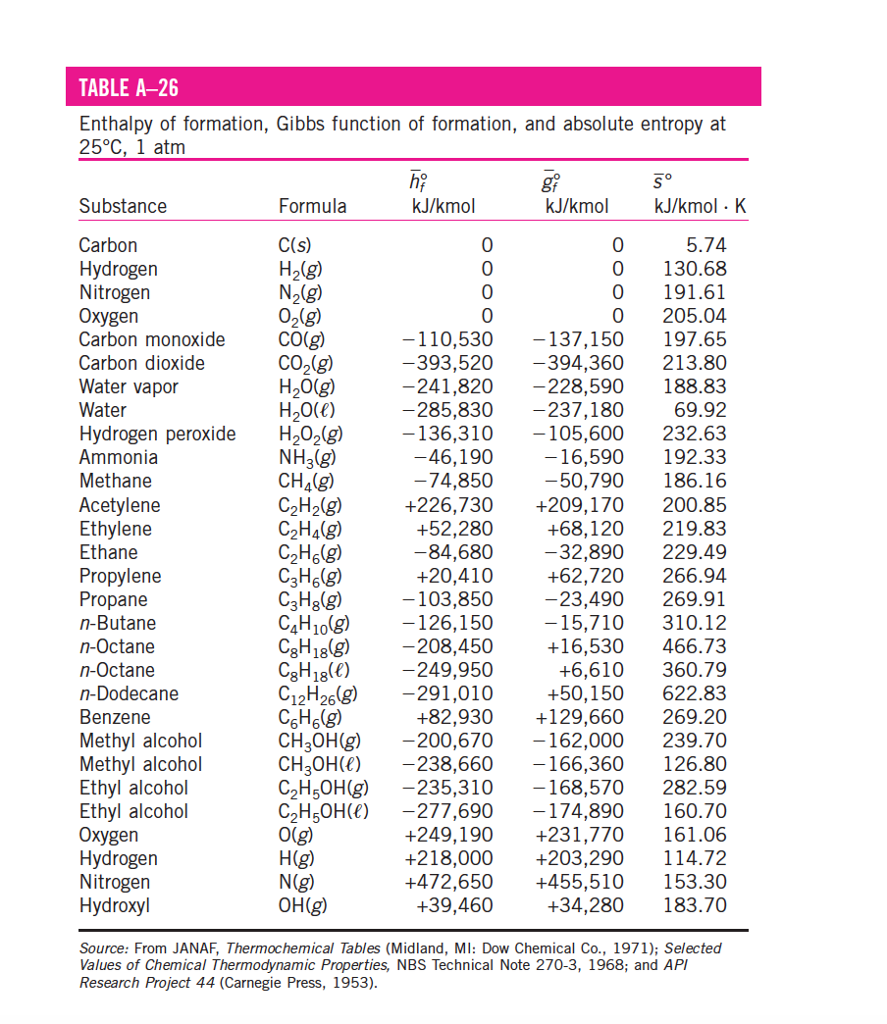
“Disorder” was the consequence, to Boltzmann, of an initial “order” not - as is obvious today - of what can only be called a “prior, lesser but still humanly-unimaginable, large number of accessible microstate Chemical structure: This structure is also available as a 2d Mol file or as a computed 3d SD file The 3d structure may be viewed using Java or Javascript. it was his surprisingly simplistic conclusion: if the final state is random, the initial system must have been the opposite, i.e., ordered. IUPAC Standard InChIKey:XLYOFNOQVPJJNP-UHFFFAOYSA-N.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
